On the morning of December 14th, 1799, George Washington was not abandoned by medicine. He was surrounded by it. His physicians bled him repeatedly, administered emetics, enemas and blistering agents, and pursued the most active therapeutics that elite eighteenth-century medicine could offer. By evening he was dead, after probable acute bacterial epiglottitis and after losing a catastrophic volume of blood. The tragedy is not that his doctors did nothing. The tragedy is that they did far too much, with confidence unrestrained by evidence.1,2
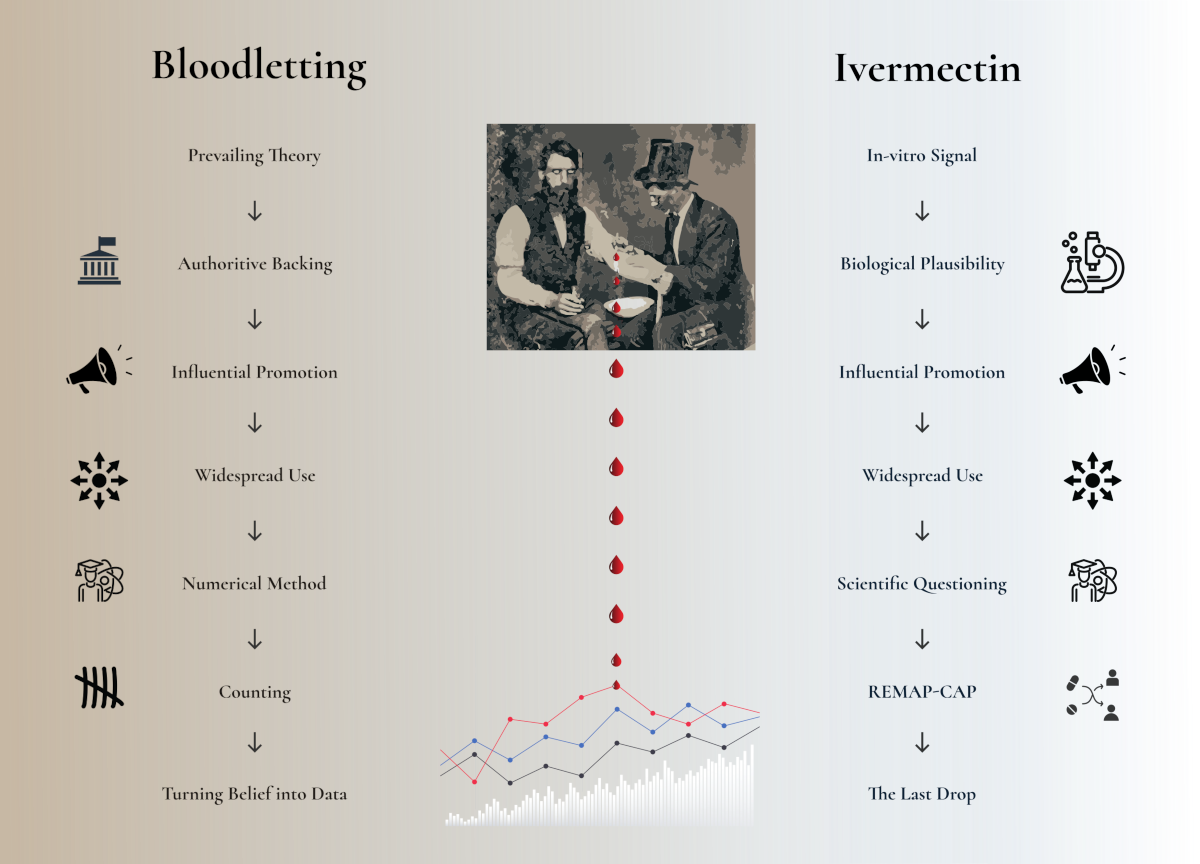
Bloodletting survived for millennia because it made sense within the dominant theory of disease. It was plausible, visible, authoritative and dramatic. It offered a grammar of action: illness was excess, imbalance, congestion; the physician restored order by opening a vein. Only when clinicians such as Pierre-Charles-Alexandre Louis began counting outcomes, comparing like with like, and asking whether bled patients actually did better did the old therapy begin to lose its scientific credibility.3,4
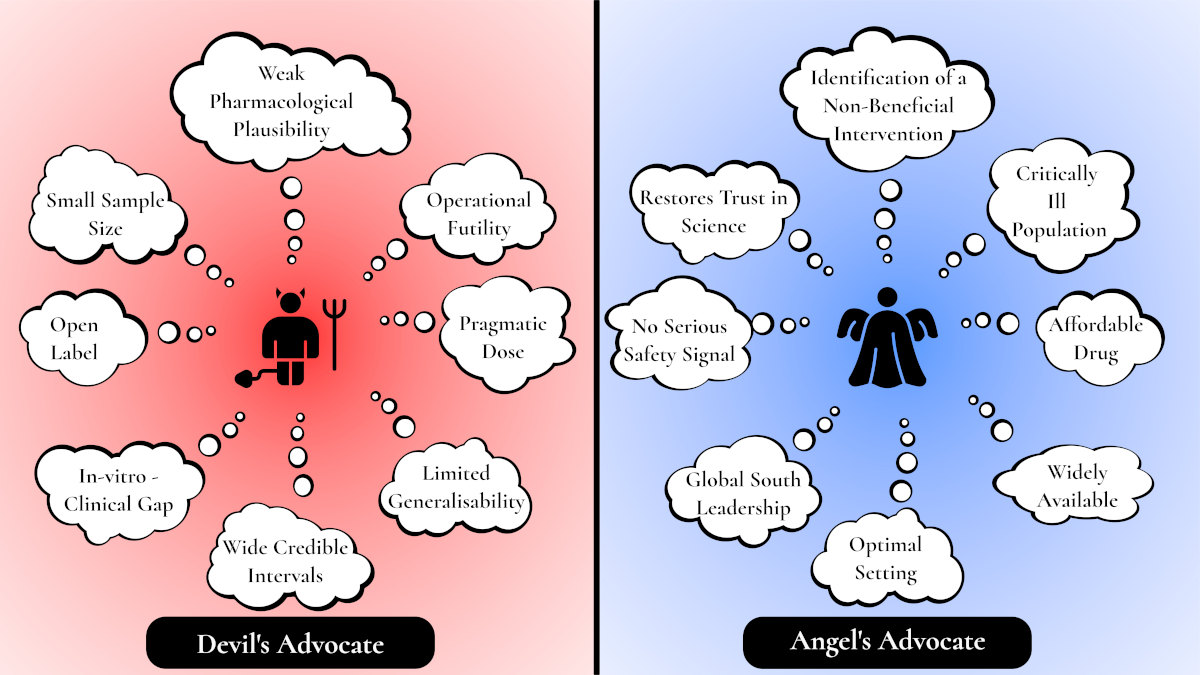
REMAP-CAP’s ivermectin trial belongs in this historical lineage. The comparison with bloodletting is not one of direct toxicity or barbarism; ivermectin, at the pragmatic dose used in REMAP-CAP, was not associated with serious adverse events. The parallel instead lies in something more enduring and more dangerous: the tendency of medicine, particularly during crisis, to confuse plausibility, confidence and interventional activity with evidence that patients actually benefit. The epistemic pattern is familiar: a biologically thin rationale, public confidence far beyond the data, influential voices promoting therapeutic certainty, and clinical use spreading before the evidence had matured. REMAP-CAP is therefore not only a trial of ivermectin. It is a modern trial of stopping.5,6
This matters because COVID-19 was not an abstract scientific inconvenience. It caused more than seven million reported deaths worldwide, while clinicians were forced to make decisions under fear, uncertainty, public scrutiny and therapeutic scarcity.7 Some treatments were implemented because randomised trials showed benefit. Others entered practice because they were inexpensive, available, plausible, emotionally attractive, or amplified by public figures whose confidence exceeded existing data. Ivermectin became the archetype of the second phenomenon.
The central lesson of this trial is therefore not simply that ivermectin was unlikely to help hospitalised patients with COVID-19. The deeper lesson is that de-implementation of a non-beneficial therapy can be as important as implementation of a beneficial one. Medicine does not progress only when it discovers a new treatment. Sometimes it progresses when it develops the discipline to put down the lancet.
Bloodletting and the seduction of doing something
Bloodletting was not a marginal practice. It was among the central rituals of pre-modern therapeutics. It crossed cultures and centuries. It was practised by physicians, surgeons and barber-surgeons. It reached its European peak in the nineteenth century, precisely at the moment when medicine was becoming more professional, not less.3 Its persistence is therefore not best explained by stupidity. It is better explained by the seductive power of a coherent but wrong model.
To the physician steeped in humoral theory, bloodletting was not irrational. Fever, inflammation, congestion and agitation all seemed to demand evacuation. The treatment was active, personalised and visible. The patient felt something. The family saw something. The physician did something. In the emotional economy of illness, that matters.
This is one reason ineffective therapies can be so hard to remove. They often satisfy needs that evidence-based medicine is bad at acknowledging: the need for agency, the need for hope, the need for a visible intervention when the honest alternative is uncertainty. Bloodletting gave medicine a ritual of action. So, in a very different way, did many pandemic repurposed therapies.
The beginning of the end of bloodletting did not come from a more elegant theory. It came from counting. Pierre-Charles-Alexandre Louis applied his “numerical method” to conditions including pneumonia, comparing outcomes among patients who were bled early and those bled later. His work was imperfect by modern standards, but conceptually radical: therapeutic authority could be challenged by grouped patient outcomes.4
That is the historical echo in REMAP-CAP. The question was not whether ivermectin had a story. It had several. The question was whether patients receiving ivermectin did better than patients who did not. The transition from story to comparison is one of the most important advances in medicine.
The confidence was never proportionate to the evidence
Ivermectin is not a fringe molecule. It is a widely used antiparasitic medicine, included on the World Health Organization Model List of Essential Medicines, with decades of experience in appropriate indications.6 That familiarity mattered. A cheap, orally available, globally accessible drug with an established safety profile is precisely the sort of intervention one would wish to work in a pandemic. If ivermectin had been effective against COVID-19, its implications for global health would have been enormous.
But wishfulness is not pharmacology. The certainty with which ivermectin was promoted was never proportionate to the quality of the evidence supporting it. The REMAP-CAP supplementary appendix is unusually explicit on this point. It describes ivermectin being promoted as a “miracle cure”, used outside trials in high-, middle- and low-income countries, advanced through advocacy groups and clinical guidance, discussed in political hearings, and associated with public narratives of dramatic recovery. It also describes lobbying, formulary pressure and even smuggling in some regions because of public demand.6
This was not a quiet drug-repurposing hypothesis awaiting evaluation. It was a social movement attached to a pharmacological claim. That distinction matters. A weak treatment hypothesis can be safely ignored when it remains marginal. Once it enters routine care, it becomes a health-system problem.
The prescribing signal was real. In the United States, outpatient ivermectin dispensing increased substantially during the pandemic, with one analysis reporting a rise from a pre-pandemic average of approximately 3,600 prescriptions per week to a peak of approximately 39,000 prescriptions in the week ending January 8, 2021.8 The CDC later warned of increased ivermectin dispensing, use of veterinary formulations, and a rise in poison control centre calls related to ivermectin exposure.9 A later national survey associated use of ivermectin or hydroxychloroquine for COVID-19 with endorsement of misinformation, lower trust in physicians or scientists, conspiracy-mindedness, and news-source patterns.10
That is the context in which REMAP-CAP should be read. The trial was not merely testing a drug. It was testing whether medicine could recover the authority of evidence after public conviction had already run ahead of it. Bloodletting had its humoral cosmology; ivermectin had its online ecosystem. In both cases, the therapy became socially useful before it was clinically proven.